
IEC 82304-1: Essential Standard for AI Healthcare Software
Explore IEC 82304-1 for Artificial Intelligence.
Learn how IEC 82304-1 provides the essential framework for AI healthcare software. Explore compliance requirements, challenges, and benefits for your organization.
The healthcare software landscape has transformed dramatically, with Software as a Medical Device (SaMD) now powering critical clinical decisions through artificial intelligence and machine learning algorithms. IEC 82304-1 provides the essential safety framework ensuring these sophisticated health software products meet rigorous international standards for patient protection and regulatory compliance.
Modern healthcare increasingly relies on intelligent software solutions that operate independently of dedicated hardware. From AI-powered diagnostic tools to machine learning algorithms predicting patient outcomes, these health software products require comprehensive safety standards. IEC 82304-1 addresses this critical need by establishing requirements for medical device software that healthcare professionals and patients can trust. This international standard ensures that health software products maintain safety and security standards equivalent to traditional medical devices while accommodating the unique characteristics of software-based solutions.
Background and Purpose of IEC 82304-1
IEC 82304-1: Health software — Part 1: General requirements for product safety was developed by IEC Technical Committee 62 to address the growing complexity of health software products in the healthcare industry. The standard emerged from the recognition that software as a medical device presents unique challenges not fully addressed by existing medical device standards.
The primary purpose of IEC 82304-1 is to establish safety and security requirements for health software products while providing a framework for software lifecycle processes specific to healthcare. It supports regulatory compliance for manufacturers and bridges the gap between traditional medical device standards and modern software development practices. According to the International Organization for Standardization, the standard applies to the safety and security of health software products designed to operate on general computing platforms and intended to be placed on the market without dedicated hardware.
Key Definitions and Concepts
Understanding IEC 82304-1 requires familiarity with several critical concepts that define the digital health landscape:
- Health Software: This refers to software intended specifically for managing, maintaining, or improving the health of individuals, or for the delivery of care.
- Software as a Medical Device (SaMD): Software used for medical purposes that is not part of a hardware medical device.
- Software Lifecycle Process: A systematic approach encompassing planning, development, verification, validation, and post-production activities.
- Product Standard: Unlike process standards that define how work is performed, this specifies requirements for the particular products themselves.
Importance of IEC 82304-1
IEC 82304-1 is critical because it formally differentiates Software as a Medical Device (SaMD) from traditional, hardware-bound medical devices. While traditional devices are assessed based on physical properties, health software presents unique characteristics that require a digital-first perspective. For instance, performance metrics shift from physical measurements to calculation accuracy and response times, while update capabilities allow for rapid modifications that necessitate specialized change management. Furthermore, the platform independence of modern software allows it to run across various computing environments, and its data-driven functionality means that overall safety is heavily dependent on algorithmic integrity and data quality.
To mitigate the specific challenges of SaMD, IEC 82304-1 provides a structured methodology focused on high-stakes reliability. It mandates comprehensive risk management by identifying software-specific hazards and implementing controls to prevent digital failure. Validation requirements ensure the software performs correctly within its intended real-world environment, while rigorous documentation standards provide the clear instructions necessary for safe operation by clinicians and patients. Finally, the standard's focus on lifecycle management supports the continuous, safe evolution of health software products as they undergo updates and improvements.

IEC 82304-1 Framework
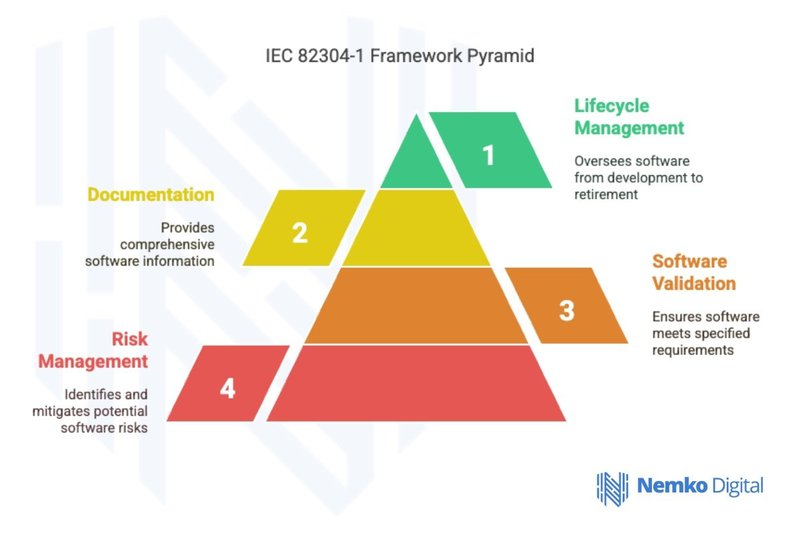
Software Validation
Software validation under IEC 82304-1 requires demonstrating that health software meets user needs and intended uses. This includes clinical validation to confirm performance in actual healthcare settings and usability validation to ensure healthcare professionals can operate the software without error. Furthermore, performance validation verifies that the software meets technical benchmarks, while interoperability testing confirms that the product exchanges data correctly with other digital health systems.
Risk Management Practices
Software risk management under IEC 82304-1 follows ISO 14971 principles while addressing software-specific concerns. The process begins with hazard identification, recognizing risks like data corruption or logic errors, followed by risk analysis to evaluate the severity of those hazards. Manufacturers must then implement risk control measures to reduce these threats to acceptable levels, while post-production risk management ensures the system is continuously monitored for emerging vulnerabilities once it is live.
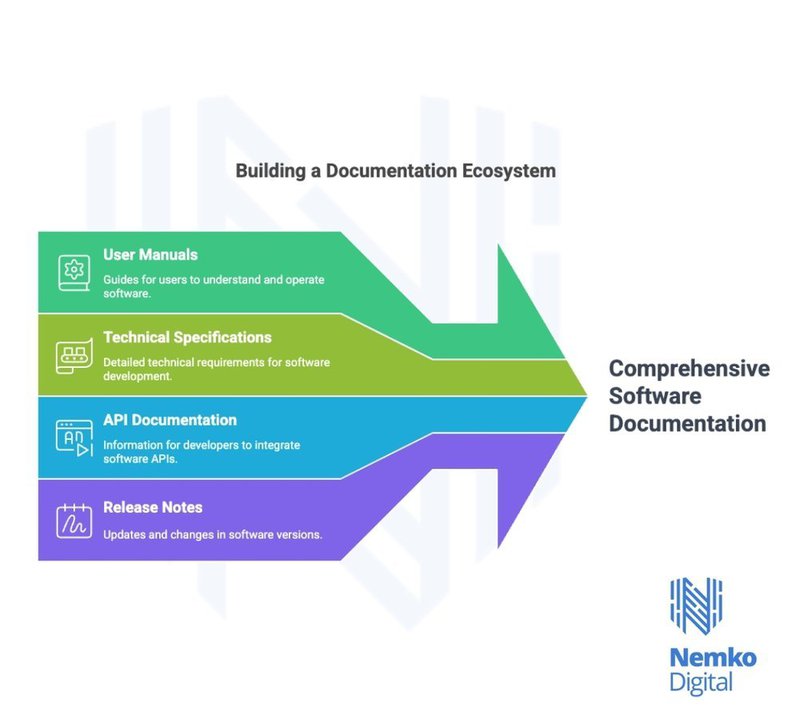
Documentation and Testing Protocols
To support safety and meet regulatory demands, the standard requires comprehensive documentation throughout the product lifecycle. This includes design specifications detailing software functionality and test documentation proving rigorous verification and validation. Furthermore, user documentation provides clinicians with clear operating instructions, while technical documentation ensures that the information necessary for maintenance and updates is always available.
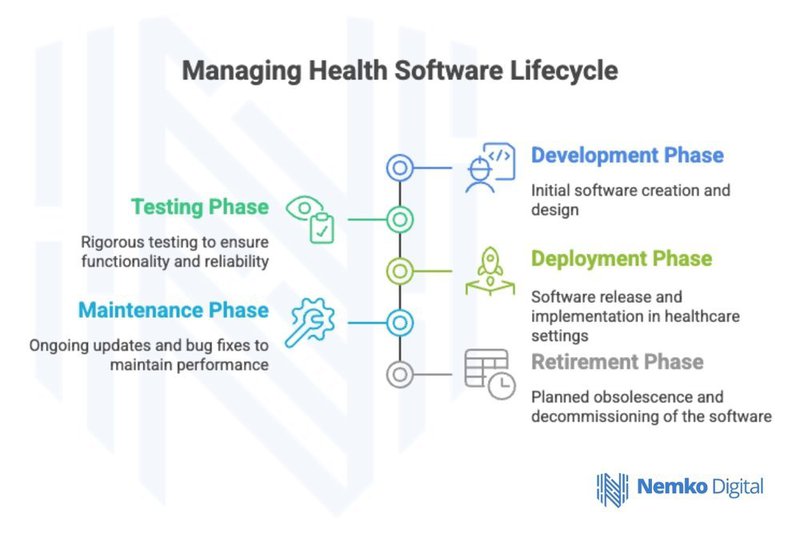
Software Life Cycle Requirements
The standard integrates seamlessly with standard IEC 62304, which defines software lifecycle processes for medical device software. This relationship ensures comprehensive coverage from initial concept through post-production management. Key lifecycle phases include:
- Planning and Requirements: Establishing clear specifications for health software functionality
- Design and Development: Implementing software architecture and algorithms
- Integration and Testing: Verifying software performance and safety
- Release and Deployment: Managing software distribution and installation
- Post-Production Management: Monitoring performance and managing updates
Implementing IEC 82304-1
Successful IEC 82304-1 implementation follows a systematic approach:
- Gap Analysis: Assess current development practices against standard requirements
- Process Enhancement: Integrate IEC 82304-1 requirements into existing workflows
- Documentation Development: Create comprehensive technical and user documentation
- Validation Planning: Design testing protocols specific to health software applications
- Risk Management Integration: Embed software-specific risk assessment throughout development
Best Practices for Adoption
Leading organizations successfully implementing IEC 82304-1 follow several key practices:
- Early Integration: Incorporating standard requirements from project inception
- Cross-Functional Teams: Engaging regulatory, clinical, and technical expertise
- Iterative Validation: Conducting ongoing testing throughout development
- User-Centered Design: Prioritizing healthcare professional and patient needs
- Continuous Improvement: Learning from post-production experience
Common Pitfalls and How to Avoid Them
Organizations should be aware of frequent implementation challenges. A primary pitfall is inadequate risk management, failing to identify software-specific hazards can lead to serious compliance gaps. Ensure comprehensive hazard analysis covering all aspects of software functionality. Additionally, insufficient documentation often creates operational risks; incomplete technical records can stall audits, making it essential to invest in thorough documentation from the project's start. Developing health software with limited clinical input is another common error, as excluding healthcare professionals often results in products that fail to meet real-world needs. Engaging clinicians early in the design process ensures the software aligns with actual medical workflows. Finally, rushing validation to meet market deadlines can lead to severe post-production problems and regulatory intervention. Inadequate validation testing can result in unforeseen issues once the software is in use, making it critical to ensure the software is proven safe and effective in its intended environment before release.
Accelerating Your IEC 82304-1 Compliance Journey
IEC 82304-1 provides the essential framework for developing safe, effective health software products in today's AI-driven healthcare environment. By understanding and implementing this standard's requirements, medical device manufacturers can navigate regulatory expectations while delivering innovative solutions that healthcare professionals and patients can trust. The standard's comprehensive approach to software lifecycle management, risk assessment, and documentation creates a solid foundation for sustainable health software development. Organizations that proactively embrace IEC 82304-1 position themselves for success in the rapidly evolving digital health landscape.
Ready to ensure your health software meets IEC 82304-1 requirements and regulatory expectations? Contact our team today to discuss how we can support your compliance journey and help you develop safer, more effective health software solutions.
Lorem ipsum dolor sit amet
Lorem Ipsum Dolor Sit Amet
Lorem ipsum odor amet, consectetuer adipiscing elit. Elementum condimentum lectus potenti eu duis magna natoque. Vivamus taciti dictumst habitasse egestas tincidunt. In vitae sollicitudin imperdiet dictumst magna.

Lorem Ipsum Dolor Sit Amet
Lorem ipsum odor amet, consectetuer adipiscing elit. Elementum condimentum lectus potenti eu duis magna natoque. Vivamus taciti dictumst habitasse egestas tincidunt. In vitae sollicitudin imperdiet dictumst magna.

Lorem Ipsum Dolor Sit Amet
Lorem ipsum odor amet, consectetuer adipiscing elit. Elementum condimentum lectus potenti eu duis magna natoque. Vivamus taciti dictumst habitasse egestas tincidunt. In vitae sollicitudin imperdiet dictumst magna.

Lorem Ipsum Dolor Sit Amet
ISO/IEC Certification Support
Drive innovation and build trust in your AI systems with ISO/IEC certifications. Nemko Digital supports your certification goals across ISO/IEC frameworks, including ISO 42001, to help you scale AI responsibly and effectively.
Contact Us
.webp)
.webp)



